Introduction: The Relatable Illusion of the Quick Fix | GLP-1 weight loss side effects
GLP-1 weight loss side effects. In the history of metabolic medicine, few developments have ignited a global frenzy comparable to the rise of glucagon-like peptide-1 receptor agonists, or GLP-1RAs. Known popularly by brand names such as Ozempic, Wegovy, and Mounjaro, these agents have moved from specialist type 2 diabetes treatments to widely promoted weight-loss solutions. The public narrative is seductively simple: a once-weekly injection that quiets intrusive food noise, slows the stomach and reduces body fat with striking efficiency.
However, as so-called Ozempic fever reaches a tipping point, a more complex clinical reality is emerging from longitudinal studies and pharmacovigilance data. While the drop on the weighing scale is real, the physiological cost of rapid weight reduction remains under-discussed in mainstream conversations. The central question is no longer whether these drugs work. They clearly do. The real question is the quality of the weight lost and what happens to the metabolic system over time.
Key Definition: What is a GLP-1 drug?
GLP-1 receptor agonists are medications that mimic the natural gut hormone glucagon-like peptide-1. They reduce appetite, slow gastric emptying, and improve blood glucose control. Drugs in this class include semaglutide, sold as Wegovy and Ozempic, and tirzepatide, sold as Mounjaro, which also targets the GIP receptor.
Takeaway 1: The “1 in 10” Experiment – A Public Health Tipping Point
The adoption of GLP-1 receptor agonists has reached a scale that materially alters the public health landscape.
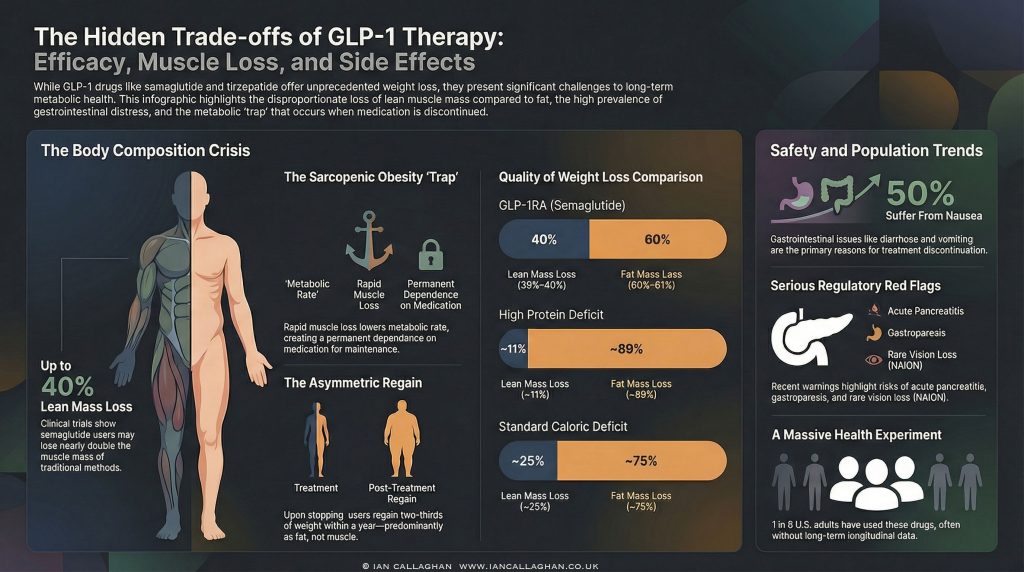
In the United States, survey and claims data from 2024 to 2025 show rapid growth. Around 12 per cent of adults reported current use by late 2025, with approximately 18 per cent reporting that they had used a GLP-1 drug at some point.
In Great Britain, usage remains lower but is rising quickly. Recent modelling suggests the broader GLP-1 economy now touches roughly one in ten adults when current use, recent use and stated intent are combined.
This represents a large real-world exposure population relative to the length of long-term safety data currently available.
US vs UK Usage Trends and Demographics
| Metric | United States (2024 to 2025) | Great Britain (2025 estimates) |
|---|---|---|
| Current usage rate | Approximately 12 percent of adults | Approximately 2.9 percent for weight loss |
| Ever used | Approximately 18 percent of adults | Approximately 4.5 percent past-year use |
| Primary demographic | Age 50 to 64 highest uptake | Age 30 to 49 strong private uptake |
| Gender pattern | Higher use in women | Majority female private prescriptions |
| Access barrier | Affordability concerns common | Lower access in deprived areas |
A further concern in the UK is the fragmentation of prescribing data. A significant proportion of weight-loss prescribing occurs through private online providers, and these prescriptions are not always fully integrated into GP records. This creates a potential pharmacovigilance blind spot for national safety monitoring.
Takeaway 2: The Gastrointestinal Reality – When “Nausea” Becomes Pathological
The most common adverse effects of GLP-1 receptor agonists are gastrointestinal. Mechanistically, this is expected because the drugs intentionally slow gastric emptying and alter gut motility.
Clinical trials often describe these effects as mild to moderate. In real-world use, a subset of patients experience significant functional impairment.
Of particular concern is drug-induced gastroparesis, where gastric emptying becomes severely delayed.
Research with liraglutide has demonstrated substantial prolongation of gastric emptying time in some individuals, with downstream risk of persistent nausea, vomiting of undigested food and reduced oral intake.
Common Gastrointestinal Side Effects
- Nausea: approximately 20 to 50 per cent
- Diarrhoea: approximately 10 to 34 per cent
- Vomiting: approximately 10 to 20per centt
- Constipation: approximately 5 to 1 per cent
- Abdominal pain: approximately 5 to per cent
- Dyspepsia or reflux: approximately 1 toper centcent
Regulators in both Europe and the UK have also added warnings regarding ileus and bowel obstruction, with frequency currently classified as unknown.
Takeaway 3: Serious Clinical Risks in the Safety Data
Beyond gastrointestinal effects, several clinically significant risks appear in regulatory labelling and post-marketing surveillance.
Clinical Red Flags
- Acute pancreatitis. A recognised class risk. UK regulators advise immediate discontinuation if suspected.
- NAION, non-arteritic anterior ischaemic optic neuropathy. A rare but potentially permanent form of vision loss is under investigation.
- Acute kidney injury. Often secondary to dehydration from persistent vomiting or diarrhoea.
- Biliary disease. Rapid weight loss increases the risk of gallstones and cholecystitis.
- Thyroid C-cell tumours. Observed in rodent models. Human relevance remains uncertain, but these drugs remain contraindicated in patients with a relevant cancer history.
- Pulmonary aspiration risk. Delayed gastric emptying increases anaesthetic risk if stomach contents are retained.
Regulatory emphasis varies slightly between regions, but these risks are present in official product information.
Takeaway 4: The Sarcopenic Risk – Losing Lean Mass
One of the most important clinical questions is body composition change, not scale weight alone.
In traditional weight loss, approximately 25 per cent of the weight lost is lean mass. GLP-1 trials show variable, and sometimes higher, lean mass loss, particularly when protein intake and resistance training are not optimised.
In the STEP 1 semaglutide trial, participants lost on average 6.9 kg of lean mass at 68 weeks. This represented roughly 39 to 40 per cent of total weight lost in that cohort.
Weight Loss Method vs Lean Mass Loss
| Method | Lean mass loss | Fat mass loss | Context |
|---|---|---|---|
| GLP-1 semaglutide | Approximately 39 to 40 percent | Approximately 60 percent | Rapid pharmacological deficit |
| Tirzepatide | Approximately 25 to 34 percent | Approximately 66 to 75 percent | Dual agonist effect |
| Bariatric surgery | Approximately 23 percent | Approximately 77 percent | Surgical intervention |
| Standard dieting | Approximately 25 percent | Approximately 75 percent | Behavioural deficit |
| High protein protocol | Approximately 11 percent | Approximately 89 percent | Muscle-preserving approach |
Loss of skeletal muscle has metabolic consequences because muscle is a major driver of resting energy expenditure and glucose disposal. In particular, among older adults, excessive loss of lean mass is associated with increased risk of frailty.
Takeaway 5: The Post-Cessation Regain Problem
Data from the STEP 1 extension study showed that after stopping semaglutide, participants regained roughly two-thirds of their prior weight loss within one year.
Body composition of regained weight varies by individual and by behaviour after cessation. However, from a physiological standpoint, fat mass typically returns more readily than lean mass unless deliberate resistance training and adequate protein intake are maintained.
Key Biological Drivers of Regain
- Ghrelin rebound, increasing hunger signals
- Leptin suppression relative to body weight
- Reduced resting energy expenditure following lean mass loss
Together, these factors create a strong biological drive toward weight regain if lifestyle structure is not in place.
Takeaway 6: The UK Access Gap
In the United Kingdom, NICE modelling suggests that millions may meet the eligibility criteria for newer agents, such as tirzepatide. However, the phased NHS rollout currently limits access to a much smaller cohort.
This has driven the rapid expansion of the private prescribing market.
Evidence suggests lower prescribing rates in more deprived regions despite higher obesity prevalence, highlighting a widening access gap.
Key Structural Pressures
- NHS capacity constraints
- Private prescribing costs
- Fragmented data reporting
- Higher entry BMI in more deprived populations
Takeaway 7: Muscle Preservation Is Not Optional
Emerging clinical consensus is clear. Weight loss without planning for muscle preservation is suboptimal.
Current Best Practice Guardrails
- Protein intake around 1.6 g per kg body weight per day
- Regular resistance training
- Monitoring of functional strength, where possible
The pharmaceutical pipeline itself reflects this concern. Combination approaches designed to preserve or increase lean mass are already under investigation, including agents targeting activin pathways.
Conclusion: A Population-Level Trade-Off?
GLP-1 receptor agonists are powerful and clinically valuable tools in appropriate populations. They are not consequence-free.
Atthe population scale, the central issue is no longer simply weight reduction. It is metabolic quality, long-term dependence, preservation of lean tissue, and the behavioural vacuum that often sits beneath pharmacological appetite suppression.
The uncomfortable reality emerging from both trial data and real-world observations is this. These drugs address the intake side of the energy equation very efficiently, but they do not automatically address the structural drivers of metabolic dysfunction.
If protein intake is inadequate, lean mass falls.
If resistance training is absent, muscle signalling drops.
If lifestyle scaffolding is weak, pressure builds the moment the pharmacology is withdrawn.
For clinicians working in obesity medicine, this is not theoretical. It is already visible in follow-up cohorts.
The Dependence Question
One of the most under-discussed implications of widespread GLP-1 use is the shift towards chronic pharmacological weight maintenance.
Unlike short-term dieting tools, GLP-1 receptor agonists exert their effects primarily through modulation of appetite and changes in gastric motility. When the drug is withdrawn, the physiological environment that supported the lower body weight often reverses.
This raises three population-level questions that remain only partially answered:
- What proportion of users will require long-term therapy to maintain weight loss?
- What is the metabolic profile of patients five to ten years after initiation?
- How will large-scale lean mass reduction in midlife populations affect frailty curves later on?
Long-term outcome data are still maturing.
The Behavioural Displacement Problem
Another emerging concern is behavioural displacement.
When appetite is pharmacologically suppressed, patients often eat less without necessarily improving dietary quality, protein distribution, micronutrient density or movement patterns. In some cohorts, this creates a situation in which weight declines while nutritional robustness does not improve in parallel.
From a purely clinical standpoint, weight loss without functional improvement is an incomplete win.
Markers that matter long term include:
- Muscle strength
- Physical function
- Protein adequacy
- Micronutrient status
- Glycaemic stability post-treatment
These are not consistently tracked within the commercial weight-loss ecosystem.
The Private Prescribing Blind Spot
In the UK, especially, the rapid expansion of private GLP-1 prescribing has created a structural monitoring problem.
When treatment is initiated outside integrated NHS pathways, several risks increase:
- Adverse events may not be fully linked to the medication in primary care records
- Medication histories may be incomplete at hospital presentation
- Long-term outcome tracking becomes fragmented
This is what pharmacovigilance specialists refer to when they discuss signal dilution in real-world data.
It does not mean the drugs are unsafe. It means the surveillance environment is imperfect and still evolving.
Who Clearly Benefits
It is equally important to state what the data does support.
In appropriately selected patients, GLP-1 receptor agonists can deliver:
- Significant weight reduction
- Improved glycaemic control
- Cardiometabolic risk improvement in defined populations
For patients with type 2 diabetes, severe obesity with complications or high cardiometabolic risk, these agents can be clinically transformative when properly supervised.
The concern raised in this analysis is not about legitimate medical use. It is about scale, context, and implementation quality in the rapidly expanding weight-loss market.
The Real Strategic Question
The global rollout of GLP-1 therapies represents one of the largest metabolic interventions in modern medicine.
The short-term efficacy story is clear.
The long-term systems story is still being written.
If lean mass preservation, resistance training, protein adequacy and structured exit planning become standard practice alongside prescribing, outcomes may remain strongly positive.
If they do not, we may see a growing cohort of patients who are lighter on the scale but metabolically more fragile than their headline weight loss suggests.
That is the trade-off now under quiet scrutiny inside obesity medicine.
The scale is moving.
The deeper question is what else is moving with it.
FAQ: GLP-1 Drugs, Weight Loss and Muscle Loss
Do GLP-1 drugs cause muscle loss?
They can. Clinical trials show that a proportion of the weight lost with GLP-1 receptor agonists is lean mass. The amount varies by individual and is strongly influenced by protein intake, resistance training, age and total calorie deficit. Without active muscle-preservation strategies, lean mass loss can be significant.
Is the weight regain after stopping GLP-1 mostly fat?
Evidence shows substantial weight regain after discontinuation in many patients. Physiologically, fat mass tends to return more readily than lean mass unless structured training and nutrition are maintained. Body composition outcomes after cessation vary widely depending on lifestyle behaviour.
Are GLP-1 drugs safe for weight loss?
For appropriately selected patients under medical supervision, GLP-1 receptor agonists can be effective and clinically appropriate. However, they carry known side effects and risks, including gastrointestinal symptoms, pancreatitis risk, gallbladder disease and rare but serious complications. They are not risk-free cosmetic tools.
Who should seriously consider GLP-1 therapy?
Current clinical guidance generally supports use in:
- Type 2 diabetes with inadequate control
- Obesity with significant comorbidities
- High cardiometabolic risk patients under supervision
Use purely for cosmetic weight loss requires careful risk-benefit consideration.
How can muscle loss be reduced on GLP-1 drugs?
Evidence-informed guardrails include:
- Adequate protein intake, commonly around 1.6 g per kg body weight
- Regular resistance training
- Monitoring of strength and functional capacity
- Avoiding excessively aggressive calorie restriction
These measures significantly influence body composition outcomes.
The Sovereign Operator Bundle gives you the full system. Three complete books plus 24/7 access to AI Ian, trained on over 40 years of real-world experience in nutrition, behaviour change and metabolic health.
Inside, you get the full EOM framework, the same principles Ian uses with clients to rebuild regulation, food control and metabolic resilience from the ground up.
This is not surface-level weight loss fluff. It is the operating manual.
Get lifetime access for £97

The Sovereign Operator Bundle: Complete Metabolic & Emotional Mastery
Stop running the same loop every bloody week.