By Ian Callaghan | Nutritional Strategist | Author of Fix Your Metabolism & The 30-Day Reset | Gut Brain Neurodegeneration
“The day I understood that neurodegeneration begins in the gut — not the brain — was the day everything changed. Not just what I ate. How I thought about ageing, about disease, about the decades I had ahead of me.”
What Nobody Is Telling You About Brain Disease and Gut Brain Neurodegeneration
Gut Brain Neurodegeneration. Here is a question your GP has almost certainly never asked you.
How’s your gut?
Not because they don’t care. But because in five-plus years of medical training, the average UK GP receives less than one week of total education on nutrition and gut health. One week. It covers the system that produces 90% of your serotonin, 50% of your dopamine, and houses more neurons than your entire spinal cord.
That blind spot is costing people their minds. Literally.
Because the science — published in leading neuroscience journals, studied at Stanford, and presented at the highest levels of academic research in 2025 — is telling us something that changes everything we thought we knew about brain disease.
Parkinson’s doesn’t start in the brain. It starts in your gut.
Alzheimer’s isn’t a memory disease. It’s a metabolic one.
Neurodegeneration isn’t inevitably bad luck. It’s a downstream consequence of a gut environment that’s been quietly failing for decades.
This is the definitive guide to understanding the gut-brain highway, how it works, and how it breaks. And what you can do, starting today, to protect your neurological future.
The Architecture of the Gut-Brain Axis: What You Were Never Taught
Your gut and your brain are in constant conversation. Not metaphorically. Physically. Through a sophisticated bidirectional communication network called the Gut-Brain Axis (GBA).
This network operates through three primary channels:
- Neural — Direct physical connection via the vagus nerve, the longest nerve in your autonomic nervous system
- Humoral — Chemical signals sent through the bloodstream, including hormones and metabolites
- Immune — The systemic movement of inflammatory cytokines and immune cells
The gut contains its own nervous system — the Enteric Nervous System (ENS) — embedded within the lining of the gastrointestinal tract. It has more than 500 million neurons. It operates with remarkable autonomy. Scientists call it the Second Brain.
And here is the architectural detail that most people miss entirely.
The vagus nerve — the physical highway connecting your gut to your brainstem — is composed of 80% afferent (sensory) fibres. That means the overwhelming majority of traffic along this nerve flows upward. Bottom-up. From your gut to your brain.
Your brain is not commanding your gut. Your gut is reporting to your brain. Constantly. Every hour of every day. Based on what you feed it.
When the report is good — a diverse microbiome, a strong gut lining, and healthy SCFA production — your brain receives signals of safety, repair, and neurological protection.
When the report is bad — dysbiosis, leaky gut, bacterial endotoxins — your brain receives alarm signals that trigger inflammation, accelerate protein misfolding, and set the conditions for the slow-motion catastrophe we call neurodegeneration.
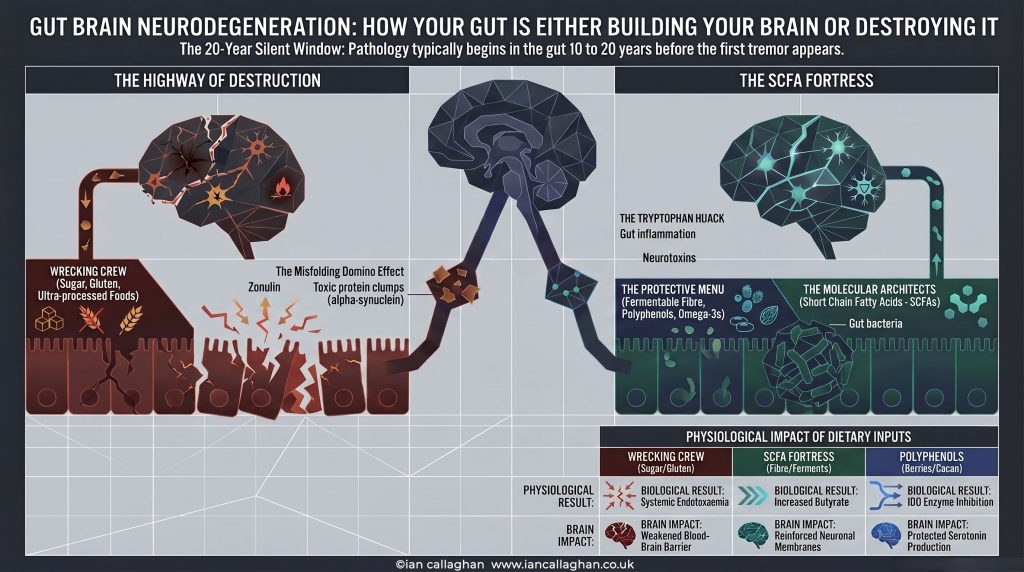
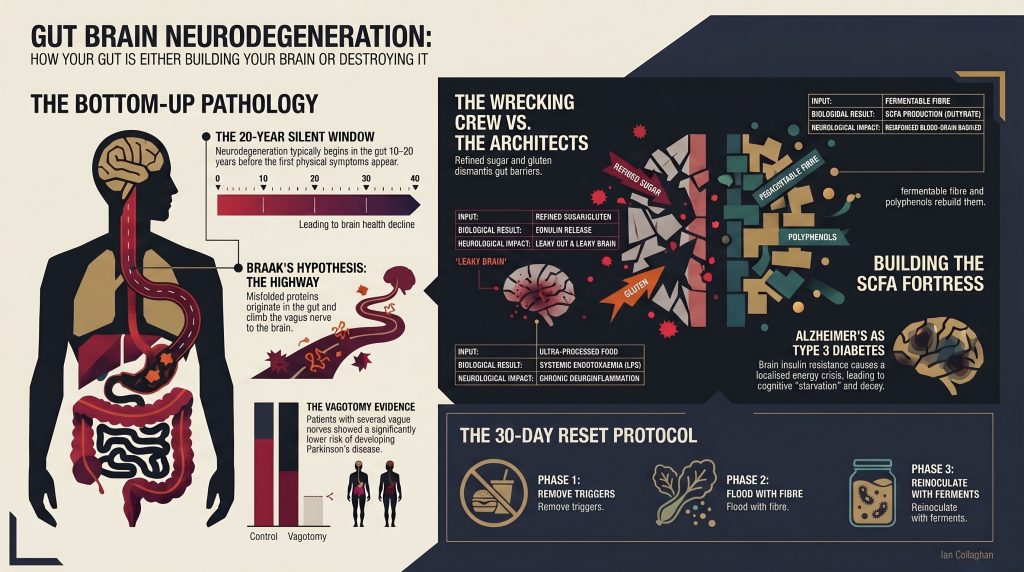
The 20-Year Silent Window: When Disease Begins Long Before Symptoms Appear
This is the part that should stop you in your tracks.
By the time a Parkinson’s patient experiences their first tremor — the shaking hand, the shuffling gait, the rigidity that changes a life overnight — the pathological process that caused it has typically been running for 10 to 20 years.
Two decades. Silent. Invisible. Building in the gut wall before the first symptom ever reaches the brain.
This is what Braak’s Hypothesis tells us. And the evidence behind it is compelling enough to have fundamentally shifted how the world’s leading neuroscientists think about disease prevention.
The implication is enormous.
If the disease starts in the gut, and the disease starts 10 to 20 years before diagnosis, then the intervention window — the period during which you can actually change the trajectory — isn’t at diagnosis. It’s right now. Decades before any neurologist ever entered the picture.
What you eat today is either building the conditions for future neurological health or quietly laying the groundwork for disease. Not as a metaphor. As a biological mechanism.
Braak’s Hypothesis: The Science That Changes Everything
In 2003, German neuroanatomist Heiko Braak proposed something that the medical establishment largely ignored for years. He suggested that Parkinson’s disease doesn’t originate in the substantia nigra — the brain region whose dopamine-producing neurons are destroyed in the disease — but in the gut’s enteric nervous system.
The mechanism he proposed goes like this.
Step 1 — The Trigger in the Gut
Environmental triggers — pesticides, bacterial toxins called lipopolysaccharides (LPS), heavy metals — breach a compromised gut lining and enter the enteric nervous system. Here, they cause a protein called alpha-synuclein to misfold. In its healthy state, alpha-synuclein is soluble and harmless. Under inflammatory stress, it changes shape. It becomes sticky. It aggregates into toxic clumps.
Step 2 — The Prion-Like Spread
These misfolded protein clumps don’t stay put. They behave almost like prions — corrupting healthy proteins around them in a domino effect. Cell by cell, the misfolding spreads through the enteric nervous system.
Step 3 — The Highway
Then the clamps find the vagus nerve. And they climb. Via a process of retrograde transport, misfolded alpha-synuclein seeds travel upward along the vagal fibres — from the gut, through the brainstem, into the midbrain, and eventually to the cortex.
The first port of call is the dorsal motor nucleus of the brainstem. This correlates precisely with the earliest non-motor symptoms of Parkinson’s — sleep disturbances, depression, constipation, loss of smell — symptoms that often appear years before the tremor.
Eventually, the seeds reach the substantia nigra. Dopamine-producing neurons die. The motor symptoms begin.
By the time you see the tremor, you’re looking at the end of a very long story that started in the gut.
The Vagotomy Evidence: The Accidental Human Trial
Here is where the science becomes genuinely jaw-dropping.
In the mid-20th century, a common treatment for stomach ulcers was a procedure called a truncal vagotomy — surgically severing the vagus nerve to reduce stomach acid production. It was performed on hundreds of thousands of patients across multiple countries over several decades.
Epidemiological data later revealed something nobody was looking for.
Patients who had undergone a full truncal vagotomy showed a significantly reduced risk of developing Parkinson’s disease later in life.
Cut the highway between the gut and the brain. Parkinson’s risk drops.
This wasn’t a small study. This was large-scale population data. And what it tells us is that the vagus nerve isn’t just a communication channel between gut and brain. In a diseased gut environment, it becomes the anatomical bridge that allows pathology to travel from one to the other.
The medical establishment had accidentally run a massive human trial. And the result pointed directly at the gut as the origin of the disease.
This evidence is now considered among the strongest support for Braak’s Hypothesis. And it has profound implications for how we think about prevention.
Related reading: The Vagal Brake: Understanding Your Autonomic Regulator | The Vagus Nerve and Mental Health
Alzheimer’s: The Type 3 Diabetes Nobody Told You About
Parkinson’s isn’t the only neurodegenerative condition being reframed through the lens of metabolic and gut health.
Alzheimer’s disease — which affects over 900,000 people in the UK and is projected to rise to 1.6 million by 2040 — is increasingly understood through a framework called Type 3 Diabetes.
This isn’t fringe science. It’s the emerging consensus of nutritional neuroscience. And it changes everything about how we think about cognitive decline.
Here’s the mechanism.
Brain Insulin Resistance
Insulin isn’t just for managing blood sugar. In the brain, insulin is required to activate glucose transporters — specifically GLUT4 — in memory-critical regions like the hippocampus. When those regions become insulin resistant, they enter a state of localised energy crisis. ATP production drops. Mitochondria fail. Synaptic connections degrade.
The brain is starving. Not of calories. Of the ability to use the energy that’s already there.
The Sugar Rust — AGEs
Chronic hyperglycaemia produces a compound called methylglyoxal, the precursor to Advanced Glycation End-products (AGEs). These compounds cross-link with brain proteins, rendering them dysfunctional and triggering a destructive cycle of chronic neuroinflammation mediated by the brain’s immune cells — microglia.
Think of it as rust. Forming slowly. Inside your brain. Over years of elevated blood sugar.
The Overwhelmed Cleanup Crew
The Insulin-Degrading Enzyme (IDE) has two jobs. It clears excess insulin. And it clears amyloid-beta — the protein associated with Alzheimer’s plaques.
In a state of chronic hyperinsulinaemia, IDE is monopolised by excess insulin. It can’t do both jobs. Amyloid-beta accumulates freely.
The brain’s cleanup crew has been overwhelmed. Not by genetics. By metabolic dysfunction driven by decades of the wrong dietary inputs.
The Methionine-Acetylcholine Connection
There’s another mechanism connecting gut health to Alzheimer’s that rarely gets discussed. Your gut microbiome modulates the metabolism of methionine, an essential amino acid. Methionine is a precursor to SAM (S-adenosylmethionine), which is required for the synthesis of choline. And choline is the precursor for acetylcholine — the neurotransmitter most associated with memory and cognitive function.
Reduced acetylcholine is a primary hallmark of Alzheimer’s disease.
Gut dysbiosis disrupts this entire pathway. It doesn’t just affect your mood or your digestion. It starves your brain of the raw materials it needs for memory.
Related reading: Your Second Brain: How Your Gut Controls Your Mood | Gut Brain Axis
The Molecular Architecture of Brain Protection: The SCFA Fortress
So what stands between your gut and neurodegeneration?
Short-Chain Fatty Acids. SCFAs.
Specifically, the trinity of butyrate, propionate, and acetate — metabolites produced by your gut bacteria when they ferment dietary fibre. These aren’t just digestive byproducts. They are your brain’s primary architects of defence.
Here’s what they actually do.
Sealing the Gut Lining
SCFAs upregulate tight junction proteins — specifically occludin — in the gut epithelium. These proteins act as molecular bouncers, sealing the gaps between intestinal cells and preventing bacterial endotoxins from leaking into the bloodstream.
No leak. No LPS. No systemic endotoxaemia. No compromised blood-brain barrier.
Reinforcing the Blood-Brain Barrier
SCFAs don’t stop at the gut wall. They enter the systemic circulation and travel to the blood-brain barrier, where they bind to G protein-coupled receptors (GPR41/GPR43) on endothelial cells, signalling them to maintain structural integrity. They upregulate occludin here, too — specifically in the frontal cortex and hippocampus.
The fortress has two walls. SCFAs build and maintain both.
Activating the Master Antioxidant Defence
Propionate specifically activates the Nrf2 pathway — the body’s master antioxidant defence system. This armours the blood-brain barrier against oxidative erosion and reduces the expression of inflammatory receptors (CD14) that would otherwise trigger neuroinflammatory cascades.
Quenching Neuroinflammation
Once in the central nervous system, SCFAs block master inflammatory pathways — specifically NF-κB and MAPK. This calms microglial activation. In animal models, it has been shown to halt the degeneration of dopaminergic neurons and preserve motor function.
The SCFA fortress is not a nice-to-have. It is the primary molecular mechanism standing between your gut environment and your neurological future.
When SCFA production collapses — as it does on a modern Western diet low in fermentable fibre — both walls come down simultaneously.
The Tryptophan Hijack: When Your Gut Turns Against Your Brain
There’s another mechanism connecting gut inflammation to neurological damage that almost nobody is talking about.
Your gut microbiome controls the fate of tryptophan — the essential amino acid required for serotonin synthesis.
Under healthy conditions, dietary tryptophan is converted to serotonin (90-95% of which is produced in the gut) or 5-HTP, which crosses the blood-brain barrier for central nervous system serotonin production.
This is your mood, your sleep, your emotional resilience. All starting in your gut.
But when the gut is inflamed, a catastrophic diversion occurs.
Pro-inflammatory cytokines — TNF-α, IL-6, IFN-γ — activate an enzyme called IDO (indoleamine 2,3-dioxygenase). This enzyme hijacks tryptophan away from the serotonin pathway and diverts it into the kynurenine pathway instead.
The end products of this diversion are neurotoxic.
- Quinolinic Acid (QUIN) — an excitotoxin that overstimulates NMDA receptors, triggering a calcium influx that destroys neuronal membranes
- 3-Hydroxykynurenine (3-HK) — generates free radicals that promote neuronal apoptosis (cell death)
Your inflamed gut isn’t just failing to produce serotonin. It’s actively producing brain-damaging toxins from the same raw material that should have become your primary mood neurotransmitter.
This is the tryptophan hijack. And it runs silently in millions of people with chronic gut inflammation — most of whom have been told their depression, anxiety, or brain fog is a mental health issue, not a gut issue.
Related reading: Healthy Gut, Healthy Mind | Gut Health, Sugar Cravings and the Microbiome
The Leaky Gut Pipeline: How Dysbiosis Becomes Neurodegeneration
Everything described above depends on one critical vulnerability: intestinal permeability—the leaky gut.
In a healthy gut, the epithelial lining is a masterwork of biological engineering. A single layer of cells secured by tight junction proteins — occludin and claudin — that act as molecular bouncers. Nutrients in. Pathogens out.
When the microbiome shifts into dysbiosis — an imbalance caused by processed foods, refined sugar, antibiotics, alcohol, and chronic stress — several things happen simultaneously.
Beneficial bacteria that produce mucins (the protective mucus layer) decline. The gut lining becomes more permeable. Gram-negative bacteria produce lipopolysaccharides (LPS) — bacterial endotoxins that can now breach the gut wall and enter the bloodstream.
This is systemic endotoxaemia. And it creates a vicious cycle.
LPS in the bloodstream triggers systemic inflammation. That inflammation weakens the blood-brain barrier. LPS enters the brain and activates microglia via the TLR4/MyD88/NF-κB pathway, thereby driving a self-sustaining cycle of neuroinflammation. The NLRP3 inflammasome — a multi-protein complex that initiates inflammatory cell death — is activated. Dopaminergic neurons die.
The leaky gut pipeline doesn’t just connect your gut to your brain. It is the mechanism by which a damaged microbiome becomes a damaged nervous system over time.
The gut is the source. The vagus nerve is the highway. The brain is the destination.
The Wrecking Crew vs The Fortress: What You’re Feeding Every Day
Your daily dietary choices are either to build the SCFA fortress or to send in the wrecking crew.
Here’s what each side looks like.
🔴 The Wrecking Crew
Refined Sugar and Processed Carbohydrates Drive hyperglycaemia and hyperinsulinaemia. Monopolise IDE. Allow amyloid-beta to accumulate—fuel pro-inflammatory bacterial species. Collapse SCFA production.
Gluten triggers the release of zonulin — a protein that physically disassembles the tight junction proteins of the gut lining. Leaky gut by design. This is the mechanism. It is not a wellness industry myth.
Ultra-Processed Food Strips the diet of fermentable fibre — the substrate your gut bacteria need to produce SCFAs. Without fibre, the architects have no materials. The fortress crumbles.
Alcohol directly damages the gut lining, disrupts the microbiome, drives LPS translocation, and compromises the blood-brain barrier. The gut-brain connection in alcohol recovery is a subject I’ve written about extensively — because it’s personal, and because the science is unambiguous.
Related reading: Gut Brain Axis in Alcohol Recovery
🟢 The SCFA Fortress
Fermentable Fibre Vegetables, legumes, oats, resistant starch. This is the raw material for butyrate-producing bacteria — Faecalibacterium prausnitzii, Roseburia intestinalis. No fibre, no SCFAs. No SCFAs, no fortress.
Polyphenols: berries, dark chocolate, green tea, olive oil, herbs and spices. Polyphenols enhance SCFA production AND inhibit the IDO enzyme — shutting down the tryptophan hijack before it starts. They are simultaneously prebiotic fuel and neurological protection.
Omega-3 Fatty Acids: Oily fish, flaxseed, walnuts. Suppress intestinal inflammation, support SCFA-producing bacteria, and protect the serotonin pathway by keeping IDO dormant.
Fermented Foods: Sauerkraut, kimchi, kefir, live yoghurt. Increase microbiome diversity — your neurological insurance policy. Specific strains like Lactobacillus rhamnosus (JB-1 and HN001) and Bifidobacterium longum have been shown to directly activate the vagus nerve and alter GABA receptor expression in the brain.
Nutrient-Dense Proteins Provide choline (for acetylcholine and the methionine pathway) and tryptophan (for serotonin, when the IDO enzyme is kept dormant by the rest of the protocol).
Top-Down Toning: The Conscious Override
Diet — the bottom-up approach — is the foundation. But the gut-brain axis is bidirectional. And there are top-down practices that directly activate the parasympathetic nervous system and improve vagal tone, creating a feedback loop that protects both gut and brain simultaneously.
Cold Water Exposure Physically triggers the vagal dive reflex — an immediate systemic shift to parasympathetic dominance. Lower heart rate. Reduced cortisol. Calmer microglia. I use this daily. The science behind it is not optional reading for anyone serious about neurological longevity.
Related reading: Cold Water Therapy
Breathwork Acts as a manual override for the autonomic nervous system. Controlled breathing through extended exhalation directly activates vagal efferent fibres — signalling safety to the enteric nervous system, improving insulin sensitivity, and reducing the inflammatory load that drives the tryptophan hijack.
Meditation operates at a systems level to calm microglial activation. Daily meditation practice reduces the neuroinflammatory baseline that, left unchecked, would progressively damage synaptic integrity.
HRV Tracking Heart Rate Variability — specifically the RMSSD metric (Root Mean Square of Successive Differences) — gives you a direct, objective measurement of vagal tone and parasympathetic activity. It is concrete proof that your interventions are working. Track it daily. Watch it improve. Your gut-brain highway is getting stronger in real time.
FAQ: The Questions I Know You’re Already Asking
Can diet actually prevent Parkinson’s disease?
There is no randomised controlled trial proving that specific dietary interventions prevent Parkinson’s in humans. What we have is a rapidly growing body of mechanistic evidence showing that gut health directly determines the biological conditions that precede neurodegeneration — often by 10 to 20 years. The mechanism is established. The intervention window is real. The vagotomy data are real. What you choose to do with that information is your call.
My GP has never mentioned any of this. Is it fringe?
It is being studied at Stanford, King’s College London, and the Mayo Clinic. It is published in Nature Neuroscience, Gut, and the Journal of Parkinson’s Disease. It is the subject of the MAMS EJS ACT-PD trials — the most sophisticated multi-arm clinical trials in neuroscience in 2025. It is not fringe. Your GP’s curriculum hasn’t caught up. That gap is what I’m here to close.
Is it too late if I’m already in my 40s or 50s?
The gut lining regenerates completely every few days. Microbiome composition shifts measurably within weeks of dietary change. The brain has extraordinary neuroplasticity. It is never too late to start. But the earlier you start, the longer the protective period you’re creating. The 20-year silent window cuts both ways — which means the next 20 years of what you eat matter more than most people will ever be told.
What about genetics?
Genetics loads the gun. Lifestyle pulls the trigger. For the vast majority of neurodegenerative disease risk, the gut environment determines whether a genetic predisposition becomes a clinical reality. The NLRP3 inflammasome, the TLR4 pathway, the IDO enzyme — these are not genetic inevitabilities. They are biological switches that your daily choices either activate or suppress
What’s the most important thing I can do right now?
Remove the wrecking crew first—Gluten, refined sugar, ultra-processed food. The gut lining cannot begin to heal while zonulin is continuously disassembling it. Week one of the 30-Day Reset is always about clearing the field before building the fortress.
The 30-Day Reset: Building Your Neurological Fortress
The protocol is simple. The discipline is the hard part.
📅 Week 1 — Remove the Wrecking Crew gluten and refined sugar. Ultra-processed food is out. Alcohol out. Watch what happens to your gut, your sleep, your mental clarity, and your brain fog within seven days. You’ll have your first data point.
📅 Week 2 — Flood with Fibre and Polyphenols. Every meal contains plants. Colour, variety, volume. Aim for 30 different plant species across the week — research shows this level of diversity dramatically shifts microbiome composition toward SCFA-producing species. Your gut bacteria are rebuilding the fortress.
📅 Week 3 — Reinoculate with Ferments Daily sauerkraut, kimchi, or kefir. This is where microbiome diversity begins to compound. Introduce the vagal-stimulating strains. Watch your RMSSD score start to climb.
📅 Week 4 — Repair and Audit: How’s the gut? The mood? The brain fog? The energy? The clarity? You have four weeks of biological data. You don’t need me or anyone else to tell you it’s working. Your body will be telling you loud and clear.
What’s Coming Next: The Bottom-Up Thesis
I’m going to be straight with you.
Everything in this article represents the tip of a very deep research process I’m currently embedded in.
The gut-brain-neurodegeneration connection — the bottom-up thesis that your gut is either building your brain or destroying it — is the subject of a book I’m developing. It goes deeper than anything currently on the market. Into the molecular origami of protein misfolding. Into the NLRP3 inflammasome and the toxic cascade. Into the 2025 clinical landscape of disease-modifying therapies. What Braak’s Hypothesis means not just for Parkinson’s but for the full spectrum of neurodegenerative conditions.
The research is ongoing. The book is coming.
If you want to be the first to know when it lands — and to get access to the research, protocols, and thinking as it develops — make sure you’re on the list.

The 30-Day Reset: Reclaim Your Vitality, Rewire Your Mind, and Reboot Your Body (Digital Ebook)
The 30 Day Reset is not a diet. It is a complete biological overhaul for anyone who is wired, tired, and done with feeling like shite. The 30-Day Reset is a 160+ page military-grade systems reboot for the over-35s. Four pillars. Eat, sleep, move, mind. One month to strip out the industrial poison, reset your dopamine pathways, silence Bob, and rebuild the machine that’s been running on the wrong fuel for decades. Not a diet. Not a programme. A complete…
The Bottom Line
Your GP has had less than a week of training on gut and nutritional health. The system they were trained in has a blind spot the size of a motorway.
But you’ve just read the science. The vagotomy data. The Braak Hypothesis. The SCFA fortress. The tryptophan hijack. The Type 3 Diabetes framework. The 20-year silent window.
You now know more about the gut-brain connection and its role in neurodegeneration than most people will ever be told by anyone in a clinical setting.
The question is never what you know. It’s what you do with it.
Your gut is either building your brain or dismantling it. Every single day. Based on what you eat.
That’s not a scare tactic. That’s the machine. And now you know how it works.
👇 Drop a comment below. Has this changed how you think about gut health and brain disease? Has anyone in your family been affected by Parkinson’s or Alzheimer’s? I want to hear from you.
Ian Callaghan is a nutritional strategist, mindset coach, and author based in the UK. His work sits at the intersection of metabolic health, gut biology, and neurological longevity. He is the author of Fix Your Metabolism and The 30-Day Reset, and is currently deep in research for a groundbreaking new book on the bottom-up thesis of neurodegeneration.
© Ian Callaghan | iancallaghan.co.uk